Of all the irreconcilable splits in science journalism, the one concerned with what we write about and how we go about has been my bugbear. On the one hand there are journalists focused on telling stories through people. On the other are journalists like me who believe there is more to acknowledging the world and understanding how it works than those with people at the hearts of their narratives can convey.
The first group is much larger and more popular because it carries forth a powerful argument: people like to read about people. Their narratives are often more easily arresting as well as command larger audiences. This argument blared to the fore when in 2017 science journalist Cassandra Willyard wrote on The Last Word on Nothing: “… humans like stories, mostly stories about other humans. I might not be interested in gravitational waves, but I am interested in science as a process. Humanize the process, and you’ll hook me every time.”
But there are many corners of the natural universe that have nothing to do with people or the human experience. There is no science sans scientists and no journalism sans readers; my point simply is there are ways and things to understand that are equally, if not better, served by not humanising the narrative, and insisting on the latter would overlook them.
Manner of things
Take the work of Ben Feringa, Jean-Pierre Sauvage, and J. Fraser Stoddard, for instance. In the 1980s, Stoddard started off trying to improve the efficacy of a herbicide, created fun molecules called catenanes and rotaxanes, and ended up in a field called molecular electronics. Just as much as these molecules would not have existed if not for the (documented) curiosity and perseverance of Stoddard et al., his curiosity and perseverance would be meaningless without the molecule itself. Sauvage et al. then found a way to make these molecules in large quantities and today they are used to make molecular machines.
In the 1990s, Ben Feringa and his team used similar chemistry to altogether build a ‘nanocar’: a block of molecules that moved on a surface when some energy was supplied to it. Scientists have since adapted many of the techniques that came about in the course of developing these wonderful machines to other applications, including for the ever-present real-world.
But just for a moment, what if we hang back and marvel at the nanocar itself? People have in fact even organised nanocar races, with molecular cars of different designs competing against each other on minuscule tracks. These things exist, and science journalism should be concerned about them as well, maintaining a space for unfettered wonder and a curiosity of the manner of things.
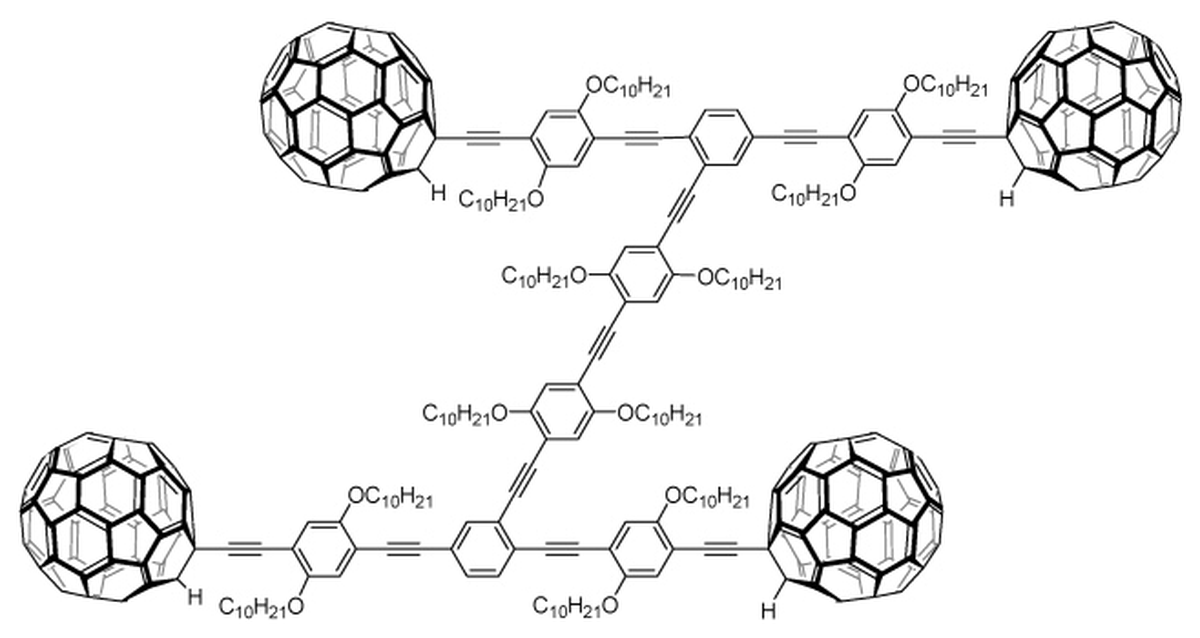
The chemical structure of a nanocar made by a team led by James Tour, reported in 2010. The team built the ‘car’ to check if fullerenes — the globes serving as the wheels — roll or slide along the surface. Shirai Y. et al.
| Photo Credit:
Shirai Y. et al.
Twisting a molecule
Consider a more recent study in the same vein, on March 5 in Science, about a molecule with a “half-Möbius topology”. Chemists have theorised for many years that molecules with unusual shapes could exist with fundamentally different properties. And they have created some of these molecules: those whose electron clouds have a Hückel topology, like a band with no twists, and with a Möbius topology, a band with a 180° twist in the middle. Now they have created a molecule whose electrons flow with a half-Möbius topology as well – in a band with a 90° twist. How did they do it?
Researchers started with a surface of salt (NaCl, the one you use at home to make tasty food), on top of which there was a ring of 13 carbon atoms plus two chlorine atoms nearby. Hovering above them all was a slender but extremely sharp needle. The researchers used the needle to draw and attach the two chlorine atoms to the ring. The ring was split into two parts: one side had six bonds and the other side had seven. When it was time to change the molecule’s shape, the researchers parked the needle directly over a specific spot and sent a small pulse of electricity down through it. Imagine the molecule was like a ball sitting in a shallow hole. To move it to a different hole, you need to give it a little push. The pulse of electricity was like this: it injected electrons into the molecule, giving it a burst of energy that sent it jumping out of its current state and into a different one.
In this new state, if the molecule stayed perfectly flat, its electrons would be unstable because they would be crowded into the same energy level. To lower its total energy, the molecule distorted itself. A chain with an even number of carbon bonds is most stable when its atoms physically twist 90° (which creates a full Möbius shape), whereas one with an odd number prefers to stay flat. By connecting two segments, one with an odd and one with an even number of bonds, the researchers forced the ring to strike a compromise. The atoms physically tilted by about 24°, which forced the path of the electrons to twist by 90°. This spiral distortion is called the helical pseudo Jahn-Teller effect. The team also found they could flip the molecule back and forth between different shapes and directions.
According to the research paper, this work could help scientists design new types of electronic parts by manipulating the way a molecule’s electrons are arranged — but here’s the thing: what if these applications never come to be? Wouldn’t this work be just as interesting? Like Stoddard’s Borromean rings (three interlocking rings of molecules that come apart even if one ring quits the arrangement) and molecular elevator (a molecule that ‘travels’ on other molecules between two floors), Feringa’s synchronous rotors, James Tour and Stephanie Chanteau’s “NanoPutians” (molecules that look like little people) or nano-trucks (like Feringa’s nanocars but which can also carry other molecules), and Philip Eaton and Leo Paquette’s molecules made solely of carbon atoms forced into the shapes of the Platonic solids (like cubane, which are hard to make because carbon-carbon bonds don’t like to bend in tight angles), we now have both fully Möbius and half-Möbius molecules.
What makes it wonderful
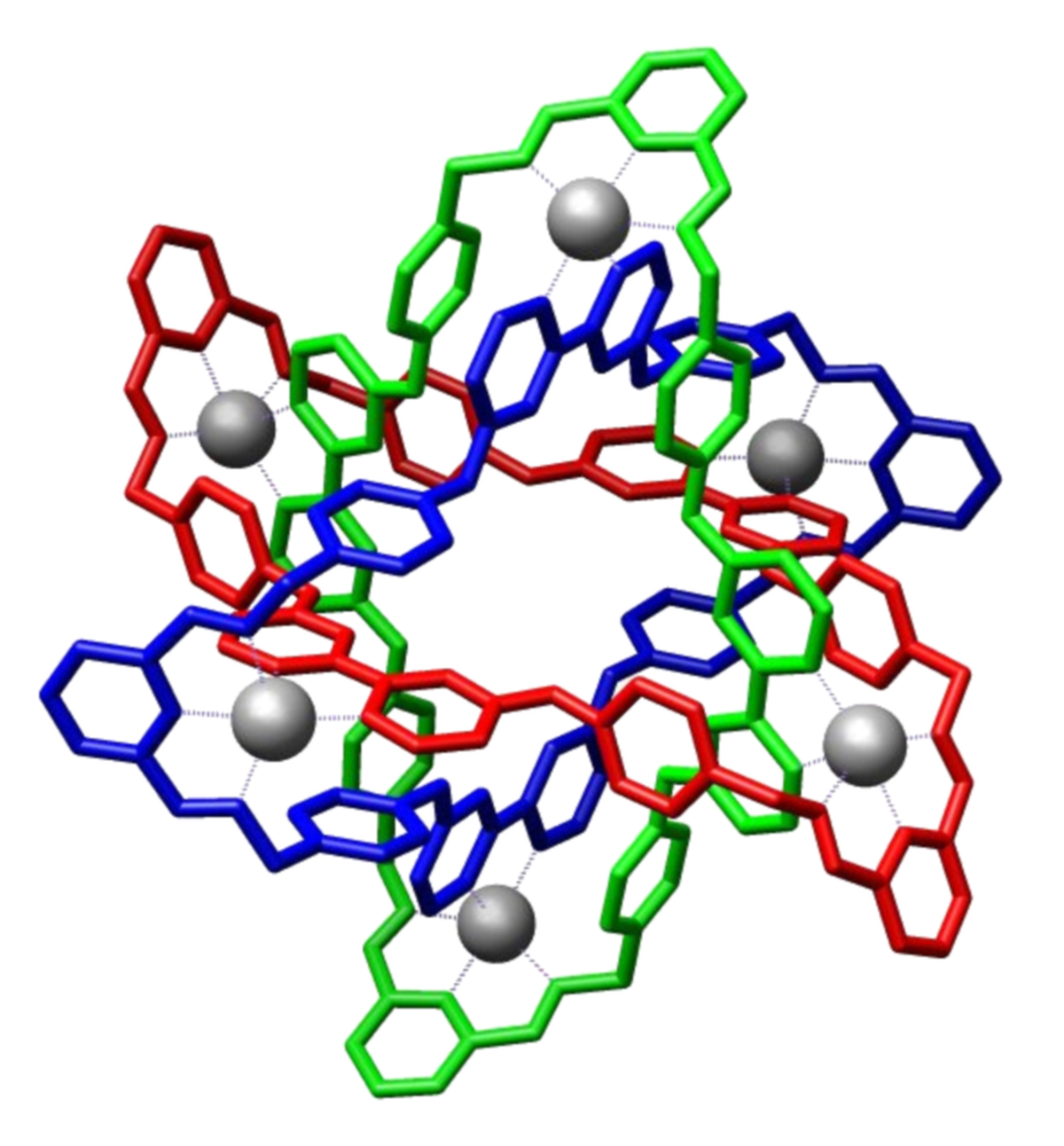
Molecular Borromean rings reported that J. Fraser Stoddart et al. reported in Science in 2004. The gray spheres are zinc ions.
| Photo Credit:
M stone (CC BY-SA)
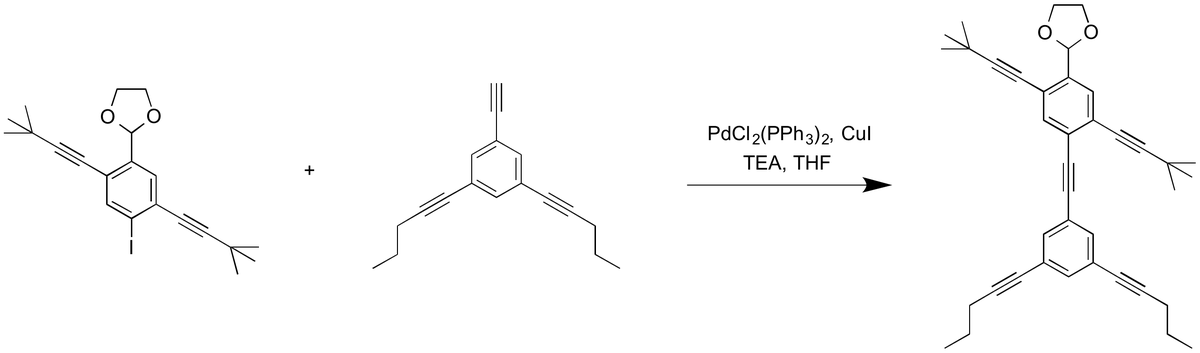
How to make a NanoPutian
| Photo Credit:
Killiannaylor (CC BY-SA)
I admit the split between the two sides is irreconcilable only in a philosophical sense: I say “look at what science allows us to do, to know”, you say “consider what the people who made these things happen went through to get there”. In the practical sense, it is easily resolved in favour of the narratives with people in them because they are just better at grabbing and holding people’s attention. But I really enjoy what this “side” allows me to do, the ideas and ‘feats’ it allows me to focus on. And I would like everyone to have that.
Granted, it’s not always fun and games like the work of Feringa, Stoddard, and Sauvage. It is often serious and often more complicated, especially for being far too removed from human experiences. But that is exactly what makes it wonderful — and what makes being able to write (or script or visualise) well enough to capture that wonder while also holding the audience’s attention a wonderful pursuit. If anything, when Willyard wrote, “You can explain gravitational waves using the cleanest, clearest, most eloquent words that exist — and you should! — but I want the story of the scientists in all their messy, human glory,” it seemed like Willyard hadn’t yet come across that one fabulous article or that we’re not doing it well enough. Or that, for reasons other than the writing, the two sides may indeed never be bridged.
mukunth.v@thehindu.co.in
Published – March 06, 2026 08:00 am IST




